Cord Blood Processing Services
Proven processing services, technology, and the assistance our clients need to grow their cord blood banking business.
FULL-SERVICE CORD BLOOD BANKING
Cord For Life® offers extensive experience, a proven processing model, superior technology, and all the assistance our clients need to grow their cord blood banking business.
Our facility is often utilized as the banking facility used by other cord blood banking brands. Virtual banks allow for national collaboration and flexibility needed for some clients.
We are experienced, technical experts who have developed a leading processing solution that is scientifically proven to deliver optimal stem cell recovery results. Cord For Life® processing techniques go above and beyond industry requirements. We do more screening/testing than our competitors.
PREMIERMAX®
PREMIERMAX® uses a powerful proprietary method for stem cell recovery, cryo-preservation, and QC sampling.
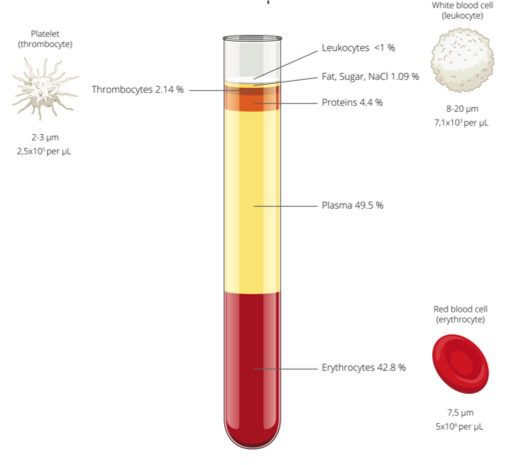
- Our process removes 98.5% of red blood cells, enabling a lower dose of free hemoglobin to recipients. Independent scientific testing proves that our process offers optimum stem cell recovery after thawing.
- Our recovery reagent is mixed with the cord blood to allow the stem cells to be separated easier, resulting in a higher amount of stem cells.
- Our preservative keeps cells from being damaged. It has less toxicity than other products currently being used by other cord blood banks.
OUR FACILITY
Our facility is utilized as the banking facility used by other cord blood banking brands. Virtual banks allow for national collaboration and flexibility needed for some clients.
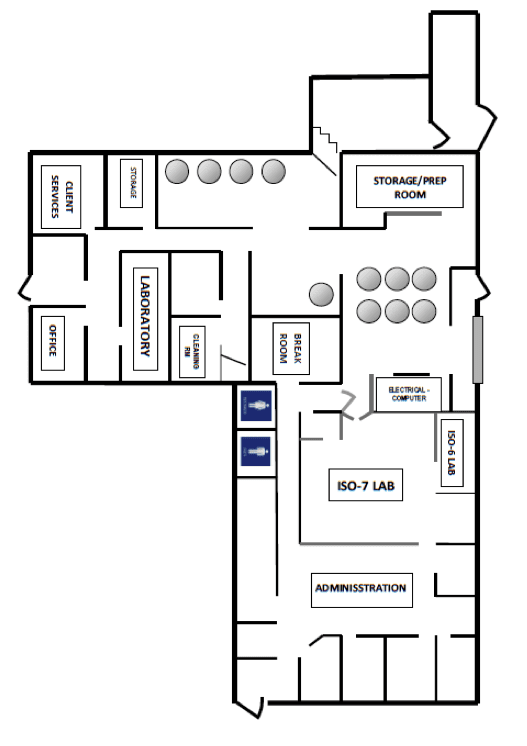
We have 7 state of the art cryopreservation storage tanks, which are capable of maintaining 40,000 umbilical cord blood stem cell units. Using liquid nitrogen vapors, these tanks are continuously monitored both manually and electronically to ensure the temperature is maintained at or below -150°C., at all times.
We can process 24 cord blood units along with cord tissue units, simultaneously per shift (3 shifts may be operated per day).
We expanded our lab in 2011 to provide 450 sq. ft. of designated ISO-7 (Class 10,000) laboratory space. In addition, it provides 120 sq. ft. ISO-6 (Class 1,000) research space allowing expansion of our alternative stem cell source process development programs.
Additionally, our Cord Tissue/BioCeutical division has 500 sq. ft. of lab space being currently renovated and satellite lab space of 120 sq. ft.
800-869-8608
info@cordforlife.com





Ready to sign-up for cord blood banking?
Reserve your Cord Blood Collection Kit in time to bring it with you when you go into labor. Give your newborn every advantage with cord blood banking!
Donate Cord Blood
By donating your newborn’s cord blood, you are joining a nationwide effort to create a genetically diverse inventory of stem cells for transplant to a child.